This informal CPD article ‘Vector Analysis in Musculoskeletal Biomechanics - Part 2: Clinical Applications and Case Interpretation’’, was provided by Dr. Mauro Lastrico, Physiotherapist at AIFiMM Formazione, an organisation recognised by the Italian Ministry of Health as an authorised CME provider. They offer organised training courses in the Mézières Method, a rehabilitative and postural approach.
Introductory Note
This article represents the third contribution in a series dedicated to applying the principles of physics to the musculoskeletal system. The first article, Clinical Assessment of Muscle Shortening [1], introduced the physical model of muscle shortening as viscoelastic deformation of the connective components, distinguishing the mechanical behaviour of the contractile component from that of the connective component. The second article, Body Equilibrium – A Physical-Clinical Interpretation of Human Upright Stability [2], presented the interpretation of human equilibrium as alignment between the weight force (G) and the ground reaction force (R), refuting the concept of "antigravity muscles".
The present article completes the theoretical framework by introducing vector analysis as a tool for identifying the muscular causes of alterations in physiological joint sequence. This is part two of a two-part article with the first part addressing Foundations and Clinical Principles.
7. Revision of the Concept of "Muscle Weakness"
Vector analysis leads to a critical revision of the concept of "muscle weakness" as a primary cause of joint alterations [4,13,23].
In traditional clinical practice, when a muscle is observed that cannot maintain a position or complete a movement, there is a tendency to conclude that the muscle is "weak" and requires strengthening [23,26]. This interpretation, apparently logical, does not consider the vectorial geometry of the forces at play [4,13].
In vectorial logic, in the absence of peripheral neurological pathologies or other pathologies that interfere with muscular contraction, subdominant actions are not impeded by the "weakness" of agonist muscles but by excess tension in dominant antagonists [4,13,23].
Referring to the shoulder example, it is not the external rotators of the humerus that are in contractile hypocapacity, but the internal rotators in excess tension (due to shortening of connective components) that impede external rotational action [20,21]. As described in the first article of this series [1], prolonged or repeated contraction, as a function of the force × time product, produces residual shortenings of connective components that determine an increase in Resistant Force (RF) at the expense of Work capacity (WF) [1,8].
7.1 The Dynamic Equilibrium Model
The dynamic equilibrium model, described in the second article of this series [2], illustrates this principle intuitively.
Imagine a central element (representing a skeletal segment) maintained in position through the co-contraction of muscular forces not symmetrically distributed [2,14]. If all forces increase the intensity of their traction, the element will move in the direction of the vectorially prevailing forces [2,14].
The forces opposing the displacement increase their tone to try to counteract it. Relative to the original position, these muscles now find themselves in a relatively lengthened position [1,8]. However, this lengthening remains within the limits of the muscle's maximum physiological lengthening capacity: this is not pathological lengthening, but different positioning within the available range [1,8].
The point is this: to return the element to central position, it is not necessary to further increase the force of the lengthened components [2,4]. This attempt would be destined to fail because the dominant forces, having geometric advantage and/or greater intensity, will continue to prevail [4,13].
The correct strategy is to create conditions so that all forces decrease their intensity, allowing the system to return towards the initial state of equilibrium [2,4]. In clinical terms: rebalancing is not achieved by strengthening muscles in lengthened position but by reducing excess tension in dominant ones [1,2,4].
7.2 Implications for Statics and Dynamics
In statics, what is perceived as "weakness" in maintaining a position is actually the effect of excess tension in dominant antagonists [2,4]. The "weak" muscle is working against elevated resistance, not against gravity or external load [2,4].
In dynamics, when both agonists and antagonists present shortenings of connective components, both muscle groups must overcome their own internal Resistant Force before producing useful movement [1,6]. This results in less fluid movements, limited in excursion, requiring greater energy expenditure or compensatory strategies to be completed [4,14].
System efficiency is compromised not because muscles are "weak" in the sense of being hypotrophic or denervated, but because a significant portion of produced energy is dissipated in overcoming internal resistances instead of producing movement [1,6,8].

8. The Role of Muscle Strengthening in the Biomechanical Model
What has been presented so far might be interpreted as a denial of the value of muscle strengthening in rehabilitation. It is essential to clarify that this is not the case [26,27].
In the biomechanical model presented here, muscle strengthening is not excluded and is not considered harmful. Simply, it does not represent the initial phase of treatment when dominant vectorial shortenings are present, because under such conditions it is unable to produce stable mechanical corrections [4,13,23].
The principle is simple and derives directly from vector analysis: a subdominant muscle cannot modify joint alignment as long as the dominant vector opposes a Resistant Force (RF) superior to the Work Force (WF) that the subdominant muscle can express [1,4].
Returning to the shoulder example: with dominant internal rotators that anteriorise the humerus due to shortening of their connective components, strengthening the external rotators cannot, by itself, reposition the humeral head [20,21]. The external rotators can certainly increase their WF, but if the RF of the internal rotators remains unchanged, the geometric equilibrium does not change [4,20].
This does not mean that strengthening is useless or incorrect, but that it requires favourable biomechanical conditions to be effective [4,26,27].
8.1 The Therapeutic Sequence: First Equilibrium, Then Strengthening
Based on the vectorial principles presented, the logical therapeutic sequence involves a preparatory phase followed by a consolidation phase [4,27].
The preparatory phase focuses on: reduction of RF in shortened muscles (through prolonged stretching techniques that act on connective components) [1,28]; rebalancing of dominant vectors; restoration of physiological joint alignment; reduction of joint conflicts and compensations [4,27].
Once coherent joint mechanics are restored, strengthening becomes fully applicable and performs essential functions [26,27]: it consolidates the correction obtained; increases the WF of previously subdominant muscles; improves functional capacity and system resilience; prevents recurrences [4,27].
In this sense, the approach described here does not oppose Exercise Therapy: it creates the conditions for strengthening protocols to work effectively [26,27].
8.2 Why This Sequence Is Necessary
Strengthening applied before vectorial rebalancing presents several critical issues [4,13,23]:
It increases the WF of the target muscle but does not modify the RF of muscles responsible for joint displacement [1,4]. If the geometry remains altered, the increase in force is dissipated in attempting to counteract dominant forces that have not been reduced.
It can increase the overall internal resistance of the system [1,8]. If shortened muscles are also stressed during strengthening exercises (which is frequent given the interconnection of muscle chains), their RF can increase further.
It tends to reinforce a compensatory pattern [4,14]. The neuromuscular system, faced with the impossibility of correcting alignment, develops alternative strategies that, if reinforced, become more stable and more difficult to modify.
In more complex cases, it can amplify mechanical conflict [20,21]. In the shoulder, for example, strengthening external rotators in the presence of shortened internal rotators can increase compressive forces on the humeral head without modifying its position.
Conversely, strengthening applied after vectorial rebalancing: stabilises recovered joint relationships; improves functional performance; reduces the probability of recurrences; enhances the stretching work done in the previous phase [4,26,27].
Operational conclusion: in the model presented here, strengthening is neither prohibited nor considered harmful, but is part of the subsequent phase, when biomechanics have been restored [4,27]. Lengthening treatment reduces RF; strengthening, applied at the appropriate time, enhances WF and consolidates functional stability. They are complementary interventions, not alternatives [1,4,26,27].
9. Active Contraction and Residual Shortening: Two Distinct Phenomena
It is essential to distinguish between the normal length reduction that occurs during active contraction and the pathological shortening of connective components [1,6,7].
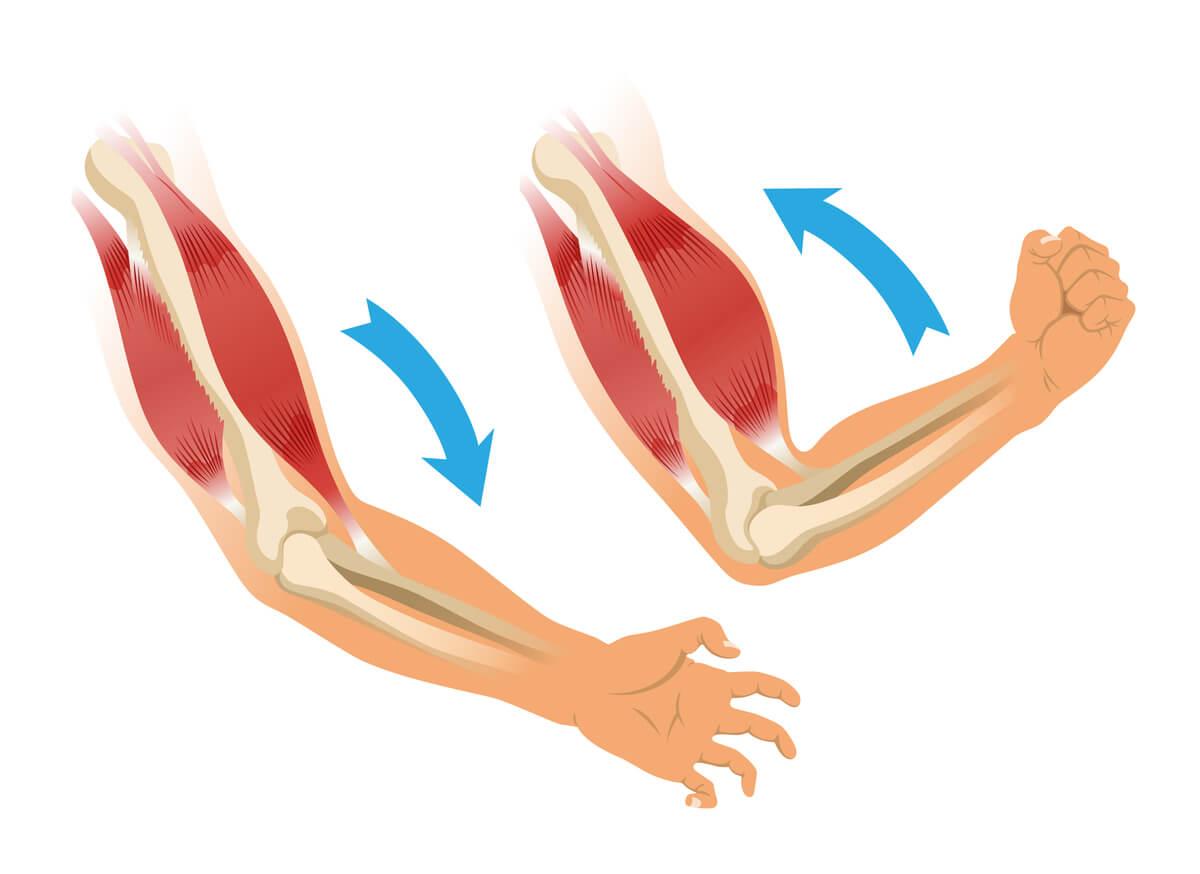
During active contraction, a muscle brings its insertions closer together, temporarily reducing its total length [6,7]. Polyarticular muscles can reduce their length by up to 20% during maximal contraction, but express greatest mechanical efficiency when shortening around 10% [6,9]. Monoarticular muscles can contract up to 50% of their resting length but maintain best mechanical efficiency between 10% and 20% of shortening [7,9].
The biceps brachii, for example, with a 20% reduction of its total length achieves complete elbow flexion, but its maximum mechanical efficiency (the point at which it generates the greatest force per unit of neural activation) occurs at mid-movement, when reduction is around 10% [6,7].
Quite different is the residual shortening of connective components, which persists even after complete muscle relaxation [1,8]. As described in the first article of this series [1], this shortening is the result of viscoelastic deformation of membranes, aponeuroses, and tendons subjected to prolonged compression during sustained contractions.
A clinically relevant finding is that minimal percentages of shortening are sufficient to significantly alter joint function: a residual shortening of 1-2% can already limit joint excursion by 10-15 degrees [1,24].
Returning to the elbow example after immobilisation, described in the first article [1]: when a cast is removed, the typical 10-15° limitation in complete extension corresponds to a residual shortening of the flexors of only 4-6 millimetres over a muscle length of about 30 centimetres, or approximately 1.5-2% [1,25].
The traditional interpretation of this situation might be: "the triceps has become weak during immobilisation and needs strengthening". Vector analysis suggests a different interpretation: it is not the triceps that is weak, but the flexors that have developed minimal residual shortening of connective components, sufficient however to prevent the last degrees of extension due to increased Resistant Force [1,8,24].
Shortening of connective components should therefore not be imagined as a macroscopically evident phenomenon: percentages of 1-2% are invisible to visual inspection but are sufficient to alter physiological joint sequence and create clinically relevant functional limitations [1,24,25].
10. Monoarticular Muscles: Active Ligaments
Monoarticular muscles deserve particular consideration in vector analysis [6,7,9].
When they develop contractile energy at maximum efficiency (approximately 10% shortening), their capacity to produce wide movement of joint segments is intrinsically limited [6,7,9]. This is not weakness but functional specialisation.
The primary function of monoarticular muscles is expressed in maintaining joint stability: they act as active ligaments capable of dynamically adapting to intra-articular stresses [9,15]. Unlike passive ligaments, they can modulate their tension in response to functional demands, increasing stability when necessary and relaxing when stability is guaranteed by other factors [9,15].
An example is represented by the piriformis muscle: considering the sacral insertion as a relatively fixed point and the trochanteric insertion as the mobile point, the main vector of the piriformis is directed towards the acetabulum [12,15]. The primary action is not external rotation (which exists as a minor vectorial component) but compaction of the femoral head into the acetabular cavity [12,15].
Vector decomposition reveals a modest abductory action and a mild external rotational action on the femur, but the main function remains stabilisation [9,12]. This explains why the piriformis, although classically described as a "hip external rotator", is not effective in producing active external rotation compared to polyarticular muscles [12].
Conclusions
Vector analysis provides the physiotherapist with a rigorous interpretive tool for identifying the muscular causes of alterations in physiological joint sequence [3,4,5]. This is not an alternative approach to evidence-based physiotherapy, but a conceptual framework that allows understanding of the mechanisms underlying observed clinical patterns [4,13].
The fundamental principles can be summarised as follows:
Muscles exert exclusively traction forces on insertions [3,6,7]. The line of force is determined by the arrangement of muscle fibres and can be represented as a vector with magnitude, direction, and sense [7,12].
Muscular forces are intrinsically asymmetric: anatomical vectorial dominances exist that, when not balanced by neural control or altered by shortenings of connective components, determine predictable patterns of joint alteration [4,13,20].
"Muscle weakness", in the absence of peripheral neurological or specific muscular pathologies, is not a primary cause but a consequence: subdominant actions are impeded by excess tension in dominant antagonists, not by contractile hypocapacity of agonists [1,4,23].
Joint rebalancing requires a logical sequence: first reduction of Resistant Force in shortened dominant muscles, then strengthening to consolidate the correction obtained [1,2,4]. Strengthening applied before vectorial rebalancing risks consolidating compensatory patterns instead of correcting them [4,13].
Minimal residual shortenings of connective components (1-2%) are sufficient to create clinically relevant functional limitations [1,24,25]. Correction of these shortenings requires prolonged mechanical stimuli that act on the viscoelastic component of connective tissue [1,8,28].
This approach, integrated with the principles presented in previous articles on muscle shortening [1] and body equilibrium [2], enables accurate and scientifically founded clinical assessment, directing therapeutic intervention towards primary causes rather than secondary manifestations [1,2,4].
We hope this article was helpful. For more information from AIFiMM Formazione, please visit their CPD Member Directory page. Alternatively, you can go to the CPD Industry Hubs for more articles, courses and events relevant to your Continuing Professional Development requirements.
References
1. Lastrico M. Clinical Assessment of Muscle Shortening. The CPD Certification Service; 2025.
2. Lastrico M. Body Equilibrium – A Physical-Clinical Interpretation of Human Upright Stability. The CPD Certification Service; 2025.
3. Winter DA. Biomechanics and Motor Control of Human Movement. 4th ed. Hoboken: Wiley; 2009.
4. Sahrmann SA. Diagnosis and Treatment of Movement Impairment Syndromes. St. Louis: Mosby; 2002.
5. Knudson D. Fundamentals of Biomechanics. 2nd ed. New York: Springer; 2007.
6. Zajac FE. Muscle and tendon: properties, models, scaling, and application to biomechanics and motor control. Crit Rev Biomed Eng. 1989;17(4):359-411.
7. Lieber RL, Ward SR. Skeletal muscle design to meet functional demands. Philos Trans R Soc Lond B Biol Sci. 2011;366(1570):1466-1476.
8. Fung YC. Biomechanics: Mechanical Properties of Living Tissues. 2nd ed. New York: Springer; 1993.
9. Herzog W. Skeletal muscle mechanics: questions, problems and possible solutions. J Neuroeng Rehabil. 2017;14(1):98.
10. Halliday D, Resnick R, Walker J. Fundamentals of Physics. 10th ed. Hoboken: Wiley; 2013.
11. Tipler PA, Mosca G. Physics for Scientists and Engineers. 6th ed. New York: W. H. Freeman; 2008.
12. Lieber RL, Fridén J. Functional and clinical significance of skeletal muscle architecture. Muscle Nerve. 2000;23(11):1647-1666.
13. Page P, Frank CC, Lardner R. Assessment and Treatment of Muscle Imbalance: The Janda Approach. Champaign: Human Kinetics; 2010.
14. Shumway-Cook A, Woollacott M. Motor Control: Translating Research into Clinical Practice. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2012.
15. Panjabi MM. The stabilizing system of the spine. Part I. Function, dysfunction, adaptation, and enhancement. J Spinal Disord. 1992;5(4):383-389.
16. Cools AM, Declercq GA, Cambier DC, Mahieu NN, Witvrouw EE. Trapezius activity and intramuscular balance during isokinetic exercise in overhead athletes with impingement symptoms. Scand J Med Sci Sports. 2007;17(1):25-33.
17. McGill SM. Low Back Disorders: Evidence-Based Prevention and Rehabilitation. 3rd ed. Champaign: Human Kinetics; 2016.
18. Adams MA, Roughley PJ. What is intervertebral disc degeneration, and what causes it? Spine. 2006;31(18):2151-2161.
19. Cholewicki J, McGill SM. Mechanical stability of the in vivo lumbar spine: implications for injury and chronic low back pain. Clin Biomech. 1996;11(1):1-15.
20. Michener LA, McClure PW, Karduna AR. Anatomical and biomechanical mechanisms of subacromial impingement syndrome. Clin Biomech. 2003;18(5):369-379.
21. Ludewig PM, Cook TM. Alterations in shoulder kinematics and associated muscle activity in people with symptoms of shoulder impingement. Phys Ther. 2000;80(3):276-291.
22. Gracies JM. Pathophysiology of spastic paresis. I: Paresis and soft tissue changes. Muscle Nerve. 2005;31(5):535-551.
23. Janda V. Muscles and motor control in cervicogenic disorders: assessment and management. In: Grant R, editor. Physical Therapy of the Cervical and Thoracic Spine. New York: Churchill Livingstone; 1994. p. 195-216.
24. Gajdosik RL. Passive extensibility of skeletal muscle: review of the literature with clinical implications. Clin Biomech. 2001;16(2):87-101.
25. Trudel G, Uhthoff HK. Contractures secondary to immobility: is the restriction articular or muscular? An experimental longitudinal study in the rat knee. Arch Phys Med Rehabil. 2000;81(1):6-13.
26. Cuthbert SC, Goodheart GJ Jr. On the reliability and validity of manual muscle testing: a literature review. Chiropr Osteopat. 2007;15:4.
27. Kisner C, Colby LA, Borstad J. Therapeutic Exercise: Foundations and Techniques. 7th ed. Philadelphia: F.A. Davis; 2017.
28. Weppler CH, Magnusson SP. Increasing muscle extensibility: a matter of increasing length or modifying sensation? Phys Ther. 2010;90(3):438-449.